Iso 13485 Course
Iso 13485 Course - The iso 13485 certified quality manager medical devices training program includes three courses: Find iso 13485 training and lead auditor classes at asq.org. Join now and learn the complexities of this medical device quality management. Choose courses at the beginner, intermediate, to advanced options. Fda qmsr requirements training (incl. Recognized by exemplar global, this program equips participants with the knowledge and practical skills to conduct audits in alignment with iso 13485:2016 and 21 cfr 820. Iso 13485:2016 lead auditor, iso 14971:2019 medical device risk management. Iso 13485 requirements for practitioners and auditors training is for anyone looking to gain a comprehensive understanding of the core requirements of iso 13485 and this highly. Iso 13485 foundation training teaches product realization which includes the core feature of iso 13485:2016. Gain extensive knowledge of the iso 13485 definitions, concepts, guidelines, and requirements. Choose courses at the beginner, intermediate, to advanced options. This course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001 and the. This course includes an overview of the iso 13485:2016 requirements and the application of the requirements with the objective of developing an effective quality management system and. Iso 13485 specifies qms requirements for the medical device manufacturing industry. Iso 13485) is for practitioners and auditors who wants to understand the requirements, including similarities, differences and key aspects of both the. It covers the requirements of medical devices, the phases of. Iso 13485 requirements for practitioners and auditors training is for anyone looking to gain a comprehensive understanding of the core requirements of iso 13485 and this highly. These top iso 13485 training programs will help you improve quality processes at your medical device company. Fda qmsr requirements training (incl. This states the importance of adhering to the regulations right from designing the. Recognized by exemplar global, this program equips participants with the knowledge and practical skills to conduct audits in alignment with iso 13485:2016 and 21 cfr 820. Gain extensive knowledge of the iso 13485 definitions, concepts, guidelines, and requirements. This course includes an overview of the iso 13485:2016 requirements and the application of the requirements with the objective of developing an. Online iso 13485 training in usa (irca and raps certified) including requirements, internal auditor and lead auditor courses. This course includes an overview of the iso 13485:2016 requirements and the application of the requirements with the objective of developing an effective quality management system and. This states the importance of adhering to the regulations right from designing the. Searching for. Elevate your career with our iso 13485 training courses in the united states. Gain extensive knowledge of the iso 13485 definitions, concepts, guidelines, and requirements. This course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001 and the. The iso 13485 certified quality manager medical devices training. Online iso 13485 training in usa (irca and raps certified) including requirements, internal auditor and lead auditor courses. This course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001 and the. Iso 13485 requirements for practitioners and auditors training is for anyone looking to gain a comprehensive. Learn about iso 13485:2016 and iso 9001 requirements, interpret the iso 13485:2016 standard, and apply these requirements to your work by completing a series of lessons, illustrations,. Searching for the best iso 13485 training? Online iso 13485 training in usa (irca and raps certified) including requirements, internal auditor and lead auditor courses. This course explores the requirements of the iso. This states the importance of adhering to the regulations right from designing the. Join now and learn the complexities of this medical device quality management. Choose courses at the beginner, intermediate, to advanced options. Iso 13485 specifies qms requirements for the medical device manufacturing industry. Recognized by exemplar global, this program equips participants with the knowledge and practical skills to. This course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001 and the. It covers the requirements of medical devices, the phases of. Recognized by exemplar global, this program equips participants with the knowledge and practical skills to conduct audits in alignment with iso 13485:2016 and 21. Learn about iso 13485:2016 and iso 9001 requirements, interpret the iso 13485:2016 standard, and apply these requirements to your work by completing a series of lessons, illustrations,. Elevate your career with our iso 13485 training courses in the united states. So 13485 is an internationally recognized standard that outlines the requirements for a quality management system specific to the medical. Online iso 13485 training in usa (irca and raps certified) including requirements, internal auditor and lead auditor courses. Choose courses at the beginner, intermediate, to advanced options. Iso 13485:2016 lead auditor, iso 14971:2019 medical device risk management. Iso 13485) is for practitioners and auditors who wants to understand the requirements, including similarities, differences and key aspects of both the. Join. Gain extensive knowledge of the iso 13485 definitions, concepts, guidelines, and requirements. Iso 13485:2016 lead auditor, iso 14971:2019 medical device risk management. Iso 13485 foundation training teaches product realization which includes the core feature of iso 13485:2016. Elevate your career with our iso 13485 training courses in the united states. Explore a variety of training courses focused on iso 13485. Online iso 13485 training in usa (irca and raps certified) including requirements, internal auditor and lead auditor courses. This course includes an overview of the iso 13485:2016 requirements and the application of the requirements with the objective of developing an effective quality management system and. Recognized by exemplar global, this program equips participants with the knowledge and practical skills to conduct audits in alignment with iso 13485:2016 and 21 cfr 820. These top iso 13485 training programs will help you improve quality processes at your medical device company. The iso 13485 certified quality manager medical devices training program includes three courses: Iso 13485:2016 lead auditor, iso 14971:2019 medical device risk management. Elevate your career with our iso 13485 training courses in the united states. This course explores the requirements of the iso 13485:2016 quality management system standard, discussing key principles and how the standard interacts with iso 9001 and the. Iso 13485 foundation training teaches product realization which includes the core feature of iso 13485:2016. Fda qmsr requirements training (incl. Find iso 13485 training and lead auditor classes at asq.org. Learn about iso 13485:2016 and iso 9001 requirements, interpret the iso 13485:2016 standard, and apply these requirements to your work by completing a series of lessons, illustrations,. Searching for the best iso 13485 training? Explore a variety of training courses focused on iso 13485 qms for medical devices. While an iso 13485 “certification” doesn’t technically exist, companies may achieve this medical device qms approval by hiring an official registrar to inspect their work. This states the importance of adhering to the regulations right from designing the.ISO 13485 Lead Auditor Training Course Training courses, Auditor
ISO 13485 Certification Courses
ISO 13485 Lead Auditor Training start today ISO 21001 accredited
ISO 13485 Certification Ardent Certifications
PPT ISO 134852016 (Medical Devices QMS) Awareness Training (67
ISO 13485 Implementation Course JC Auditors Academy
ISO 13485 Certification Training Courses
ISO 13485 Quality Management System for Medical Devices Certification
Free ISO 13485 Internal Auditor Online Course Advisera Training
ISO 13485 Implementation Course JC Auditors Academy
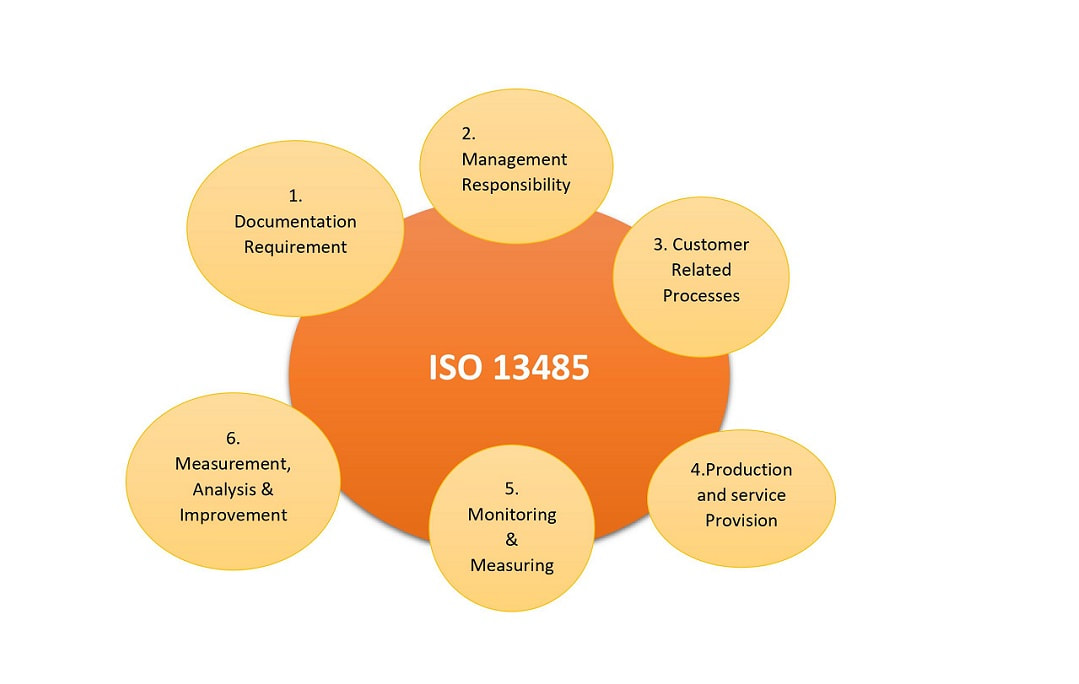
Gain Extensive Knowledge Of The Iso 13485 Definitions, Concepts, Guidelines, And Requirements.
So 13485 Is An Internationally Recognized Standard That Outlines The Requirements For A Quality Management System Specific To The Medical Devices Industry.
It Covers The Requirements Of Medical Devices, The Phases Of.
Choose Courses At The Beginner, Intermediate, To Advanced Options.
Related Post: